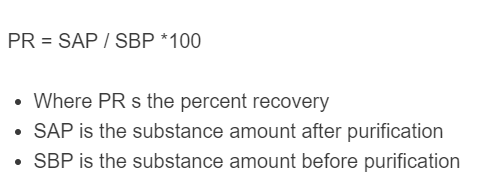Enter the total mass recovered after purification and the original starting mass to calculate percent recovery. The calculator also solves for the recovered amount or the required starting mass when the other values are known.
Percent Recovery Formula
The following equation is used to calculate the percent recovery from any purification process.
\%R = \frac{m_f}{m_i} \times 100- %R = percent recovery (%)
- m_f = final mass after purification (any unit)
- m_i = initial mass before purification (same unit as m_f)
Both masses must use the same unit. The formula applies to any physical separation process where no chemical reaction occurs.
Percent Recovery Definition
Percent recovery measures how efficiently a target substance is isolated from a mixture during a physical separation process. It applies to purification techniques such as recrystallization, liquid-liquid extraction, distillation, and chromatography, where no chemical transformation takes place. This distinguishes it from percent yield, which measures reaction efficiency in chemical synthesis where reactants are converted into new products.
A value below 100% is expected and normal. Losses occur due to residual solubility in the mother liquor, mechanical transfer between vessels, and incomplete separation. A value above 100% always indicates contamination or measurement error, not a real gain in mass.
Typical Recovery Ranges by Purification Method
Expected percent recovery varies significantly by technique. The table below shows typical ranges and the dominant loss mechanism for each method.
| Purification Method | Typical Recovery Range | Primary Loss Mechanism |
|---|---|---|
| Recrystallization | 60 to 90% | Compound dissolved in cold mother liquor |
| Liquid-liquid extraction | 70 to 95% | Unfavorable partition equilibrium |
| Column chromatography | 50 to 90% | Adsorption to stationary phase |
| Steam distillation | 50 to 80% | Incomplete condensation |
| Protein purification (HPLC) | 60 to 95% | Non-specific binding and denaturation |
Theoretical Maximum Recovery in Recrystallization
For recrystallization, the theoretical maximum percent recovery can be calculated directly from the compound's solubility at the cold filtration temperature. Some compound will always remain dissolved in the mother liquor, setting a hard ceiling on recovery that no technique improvement can overcome.
\%R_{max} = \left(1 - \frac{S_c \times V}{m_i}\right) \times 100- S_c = solubility at the cold filtration temperature (g/mL)
- V = volume of recrystallization solvent used (mL)
- m_i = initial mass of compound (g)
Example: Benzoic acid has a solubility of 0.29 g/100 mL in water at 25°C. Starting with 2.0 g dissolved in 20 mL of water, the theoretical maximum recovery is: (1 - 0.0029 x 20 / 2.0) x 100 = 97.1%. Actual recoveries are lower due to mechanical losses during filtration and transfer.
Percent Recovery Example
How to calculate percent recovery?
- Measure the initial mass before purification.
Weigh the substance before any purification step. For example, 5.00 g of crude acetanilide.
- Measure the recovered mass after purification.
After recrystallization and drying, weigh the purified product. For example, 3.80 g of purified acetanilide.
- Apply the percent recovery formula.
Divide recovered mass by initial mass and multiply by 100: (3.80 / 5.00) x 100 = 76.0%. A 76% recovery is typical for acetanilide recrystallized from hot water.
FAQ
Percent recovery is a measure of how much of a target substance was successfully isolated after a physical purification process such as recrystallization, extraction, or chromatography. It is expressed as a percentage of the original starting mass.
A recovery of 70 to 90% is generally considered good for most lab-scale purification. Values above 90% indicate a highly efficient process; values below 50% suggest significant losses or a technique issue. The acceptable range depends on the method: recrystallization typically yields 60 to 90%, while liquid-liquid extraction can reach 70 to 95%.
Yes. A result above 100% means the recovered mass exceeds the starting mass, which always signals contamination or measurement error. Common causes include residual solvent that was not fully dried from the product, co-precipitated impurities, or weighing errors. It does not represent a real gain in the target compound.
Percent yield measures how much product formed relative to the theoretical maximum in a chemical reaction. Percent recovery measures how much of an already-existing compound was isolated from a mixture, with no chemical transformation occurring. Percent yield is used in synthesis; percent recovery is used in purification and separation.