Calculate decay factors, remaining amounts, or original amounts for radioactive decay from rate, half-life, λ, time, or isotope inputs.
Decay Factor Formula
The calculator uses one of three core relationships depending on which tab you select.
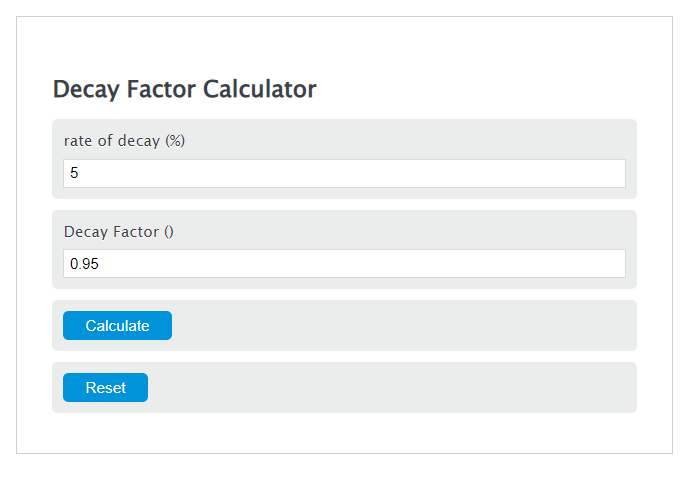
Basic mode converts a known decay rate into a decay factor:
f = 1 - r
Amount Over Time mode applies the factor across multiple periods, or uses the continuous exponential form:
N(t) = N0 * f^t = N0 * e^(-lambda * t)
Half-Life / λ mode converts between half-life, decay constant, and the per-period factor:
lambda = ln(2) / T_half f = e^(-lambda * t)
- f — decay factor (fraction remaining after one period)
- r — decay rate per period (as a decimal)
- N0 — initial amount or activity
- N(t) — amount remaining after time t
- t — elapsed time, in the same units as the period or λ
- T_half — half-life
- lambda (λ) — decay constant
The formulas assume first-order decay with a constant rate. They apply to radioactive isotopes, depreciating quantities, drug elimination at steady kinetics, and any other process where the rate of loss is proportional to the current amount. Time units must match across inputs. If you mix percent and decimal entries, the calculator handles the conversion based on the unit you pick.
The Basic tab returns f from r in one step. The Amount tab raises f to the elapsed time (or evaluates e^(-λt)) and multiplies by your starting quantity, with an option to back-solve for the original amount when you know the current value. The Half-Life tab derives λ from T_half (or vice versa) and then computes the factor for any period length you specify.
Reference Tables
Common decay rates and their matching factors:
| Decay rate per period | Decay factor (f) | Remaining after 10 periods |
|---|---|---|
| 1% | 0.99 | 90.4% |
| 5% | 0.95 | 59.9% |
| 10% | 0.90 | 34.9% |
| 20% | 0.80 | 10.7% |
| 50% | 0.50 | 0.098% |
Half-life and decay constant for common isotopes:
| Isotope | Half-life | λ | Daily factor |
|---|---|---|---|
| Tc-99m | 6 hours | 0.1155 / hr | 0.0625 |
| I-131 | 8.02 days | 0.0864 / day | 0.9172 |
| Co-60 | 5.27 years | 0.1315 / yr | 0.99964 |
| Cs-137 | 30.17 years | 0.02297 / yr | 0.999937 |
| C-14 | 5730 years | 1.21e-4 / yr | ≈ 1.000 |
Worked Examples and FAQ
Example 1: Rate to factor. A population drops 7% per year. The decay factor is 1 − 0.07 = 0.93. After 5 years, the share remaining is 0.93^5 = 0.696, or 69.6%.
Example 2: Isotope activity. An I-131 sample reads 200 mCi today. After 16 days (about two half-lives), the activity is 200 × 0.5^(16/8.02) = 200 × 0.2497 = 49.9 mCi.
Example 3: Back-solving. A Tc-99m vial reads 12 mCi at 2:00 PM and you want the activity at 8:00 AM that morning (6 hours earlier, one half-life). Original = 12 / 0.5 = 24 mCi.
Is decay factor the same as decay rate? No. The rate is the fraction lost each period. The factor is the fraction kept. They sum to 1.
When should you use λ instead of half-life? Use λ when your data comes from continuous exponential models or when the time step does not divide evenly into the half-life. The two are interchangeable through λ = ln(2) / T_half.
Can the factor be greater than 1? Not for decay. A factor above 1 describes growth, which this calculator does not model.
Why does my answer use scientific notation? Very long elapsed times produce factors that round to extremely small numbers. Scientific notation preserves the precision instead of showing zeros.