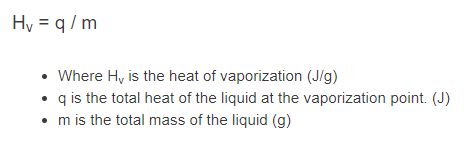Enter the heat added during vaporization (latent heat, measured while the liquid boils at the chosen pressure) and the mass that vaporized into the calculator to determine the heat of vaporization. If you also need the energy to warm the liquid up to its boiling point, use the “Heat + Boil + Vaporize” tab.
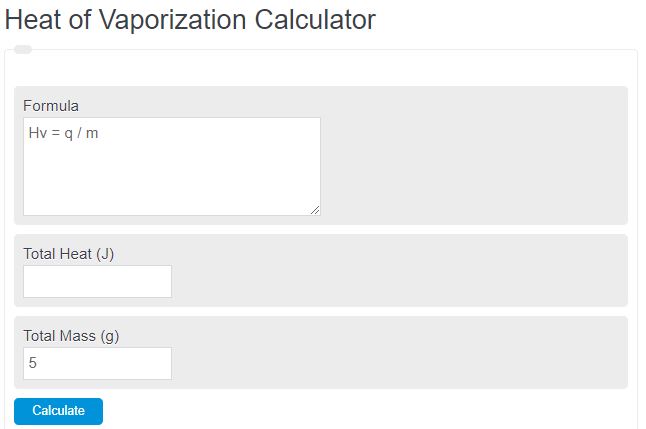
Heat of Vaporization Formula
The following equation is used to calculate the heat of vaporization (specific latent heat of vaporization).
H_v = q / m
- Where Hv is the heat of vaporization (J/g)
- q is the heat transferred to vaporize the sample (latent heat added during the phase change at the specified pressure), not the heat needed to warm the liquid up to its boiling point (J)
- m is the mass vaporized (g)
To calculate a heat of vaporization, divide the heat added during the phase change by the mass that vaporized.
Heat of Vaporization Definition
The heat of vaporization (also called the enthalpy of vaporization, when measured at constant pressure) is the amount of energy per unit mass required to convert a liquid into a vapor at constant temperature and pressure (typically at the boiling point for the chosen pressure, such as 1 atm).
Heat of Vaporization Example
How to calculate heat of vaporization?
- First, determine the mass of the liquid.
Measure the total mass of the liquid being vaporized (or the mass that actually vaporizes).
- Next, determine the heat used for vaporization.
Heat the liquid to its boiling point, then continue adding heat while it boils. Measure only the energy added during boiling (the phase change) to vaporize the measured mass.
- Finally, calculate heat of vaporization.
Divide the vaporization heat by the mass vaporized to determine the heat of vaporization.
FAQ
Heat of vaporization is the energy per unit mass required to vaporize a liquid at constant temperature and pressure (typically at its boiling point for the chosen pressure).
Vaporization is the process of turning a liquid into a gas.