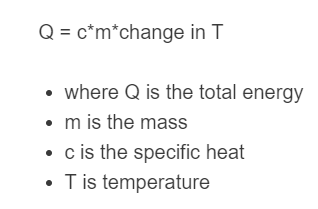Calculate energy, mass, temperature change, or specific heat from E = c × m × ΔT with selectable J, kg, °C, cal, kcal, and BTU units.
Specific Heat Capacity Guide
Specific heat capacity measures how much heat energy a material needs to change temperature. This calculator solves for the missing value when any three of the following are known: heat energy, mass, temperature change, and specific heat. Use it for single-phase heating or cooling problems where the material does not melt, freeze, boil, or condense during the process.
Core Equations
Q = c \cdot m \cdot \Delta T
c = \frac{Q}{m \cdot \Delta T}m = \frac{Q}{c \cdot \Delta T}\Delta T = \frac{Q}{c \cdot m}Variable Reference
| Symbol | Quantity | Meaning | Common Units |
|---|---|---|---|
| Q | Heat energy | Energy added to or removed from the material | J, kJ, cal, kcal, BTU |
| m | Mass | Amount of material being heated or cooled | kg, g, lb, oz |
| ΔT | Temperature change | Difference between final and initial temperature | °C, K, °F |
| c | Specific heat capacity | Material property showing energy needed per unit mass per degree | J/(kg·°C), cal/(g·°C), BTU/(lb·°F) |
How to Use the Calculator
- Enter any three known values.
- Select the correct unit for each input.
- Use the temperature change, not just the ending temperature.
- Calculate to solve for the missing quantity.
- Check that the result is reasonable for the material and temperature range.
Temperature and Unit Notes
\Delta T = T_{\text{final}} - T_{\text{initial}}\Delta T_{\mathrm{K}} = \Delta T_{^\circ \mathrm{C}}\Delta T_{^\circ \mathrm{F}} = 1.8 \cdot \Delta T_{^\circ \mathrm{C}}| Topic | Practical Reminder |
|---|---|
| Temperature difference | A change of 1 K is the same size as a change of 1°C. |
| Unit consistency | Match the mass and temperature units to the specific heat units. |
| Cooling problems | If temperature decreases, Q may be negative depending on sign convention. |
| Phase changes | If the material melts, freezes, boils, or condenses, latent heat must also be considered. |
| Gases | Specific heat can differ for constant-pressure and constant-volume conditions. |
Approximate Specific Heat Values
These values are useful for estimation. Actual specific heat varies with temperature, pressure, composition, and physical state.
| Material | Approx. Specific Heat | Notes |
|---|---|---|
| Water (liquid) | 4184 J/(kg·°C) | High heat storage; common benchmark |
| Ice | 2090 J/(kg·°C) | Much lower than liquid water |
| Steam | 2010 J/(kg·°C) | Varies with pressure and temperature |
| Air | 1005 J/(kg·°C) | Approximate constant-pressure value |
| Aluminum | 900 J/(kg·°C) | Heats faster than water, slower than many metals |
| Concrete | 880 J/(kg·°C) | Useful in building thermal calculations |
| Glass | 840 J/(kg·°C) | Typical room-temperature estimate |
| Iron / Steel | 450–500 J/(kg·°C) | Common engineering range |
| Copper | 385 J/(kg·°C) | Conducts heat well but has moderate specific heat |
| Lead | 128 J/(kg·°C) | Low specific heat; temperature changes quickly |
Quick Example
If 2 kg of water is heated by 15°C, the required heat energy is:
Q = 4184 \cdot 2 \cdot 15 = 125{,}520 \text{ J}This is 125.52 kJ of heat added to the water.
Common Mistakes
- Using the final temperature instead of the temperature change.
- Mixing unit systems without converting them.
- Using a liquid value for a solid or gas version of the same substance.
- Ignoring phase change energy near melting or boiling points.
- Assuming specific heat stays exactly constant over very large temperature ranges.
FAQ
What is the difference between specific heat and heat capacity?
Specific heat is per unit mass. Heat capacity is for the entire object.
C = m \cdot c
Why can temperature change be entered in either °C or K?
Because a temperature interval of 1 K is the same size as an interval of 1°C.
Why does water take so much energy to heat?
Water has a high specific heat capacity, so each kilogram requires a relatively large amount of energy for each degree of temperature rise.
Can this calculator be used for cooling?
Yes. The same relationship applies when heat is removed; only the sign of Q changes if you are tracking direction of heat flow.
When should this equation not be used by itself?
Do not rely on the simple form alone when phase change occurs or when specific heat changes significantly across the temperature range being analyzed.