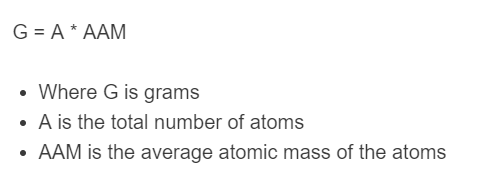Calculate atoms from grams or grams from atoms for any element using atomic mass and Avogadro’s number, including custom atomic masses.
- All Chemistry Calculators
- Average Atomic Mass Calculator
- Grams to Moles calculator
- Moles to Atoms Calculator
| Calculator Operations | Required Inputs |
|---|---|
| Convert Grams to Atoms | Grams, Average Atomic Mass |
| Calculate Molar Mass (g/mol) from Grams and Atoms | Grams, Number of Atoms |
| Convert Atoms to Grams | Number of Atoms, Average Atomic Mass |
Atoms to Grams Formula
The following equation is used to calculate the total number of grams from atoms.
G = A * AAM / (6.02214076e23)
- Where G is grams
- A is the total number of atoms
- AAM is the average atomic mass (molar mass) in g/mol
To calculate grams from atoms, multiply the number of atoms by the molar mass (g/mol), then divide by Avogadro’s number (6.02214076×10^23 atoms/mol).
| Atoms | Grams (g) |
|---|---|
| 1×10^20 | 0.00199 |
| 2×10^20 | 0.00399 |
| 5×10^20 | 0.00997 |
| 1×10^21 | 0.0199 |
| 2×10^21 | 0.0399 |
| 5×10^21 | 0.0997 |
| 1×10^22 | 0.199 |
| 2×10^22 | 0.399 |
| 5×10^22 | 0.997 |
| 1×10^23 | 1.99 |
| 2×10^23 | 3.99 |
| 5×10^23 | 9.97 |
| 6.02214076×10^23 | 12.0 |
| 1×10^24 | 19.9 |
| 1.5×10^24 | 29.9 |
| 2×10^24 | 39.9 |
| 3×10^24 | 59.8 |
| 5×10^24 | 99.7 |
| 1×10^25 | 199 |
| 1×10^26 | 1990 |
| * Rounded to 3 significant figures. Assumes Carbon (12.01 g/mol). Avogadro’s number: 6.02214076×10^23 mol⁻1. Formula: grams = atoms × (12.01 g/mol) ÷ (6.02214076×10^23). | |
Atoms to Grams Definition
Converting atoms to grams uses Avogadro’s number (6.02214076×10^23 atoms/mol) to convert atoms to moles, then uses the molar mass (g/mol) to convert moles to grams.
How to convert atoms to grams?
Example Problem:
First, determine the total number of atoms in the substance. Calculate the total number of atoms you are measuring.
For this example, the number of atoms in the sample is 6.02214076×10^23 atoms (which is 1 mole of atoms).
Next, determine the average atomic mass (molar mass) in g/mol. For more information on how to do this, you can visit the average atomic mass calculator linked above.
In this case (carbon), the molar mass is approximately 12.01 g/mol.
Finally, calculate the amount of grams in the substance using the formula above:
G = A * AAM / (6.02214076×10^23)
G = (6.02214076×10^23 * 12.01) / (6.02214076×10^23)
G = 12.01 grams
FAQ
Average atomic mass (also called atomic weight) is the weighted average of the masses of an element’s naturally occurring isotopes, using their fractional abundances.
An atom is the smallest unit of a chemical element that retains that element’s chemical properties.