Calculate microbial D-value from initial and final CFU counts, or find exposure time needed for a chosen log reduction in seconds, minutes, or hours.
- All Statistics Calculators
- Log Reduction Calculator
- Decay Constant Calculator
- Decay Factor Calculator
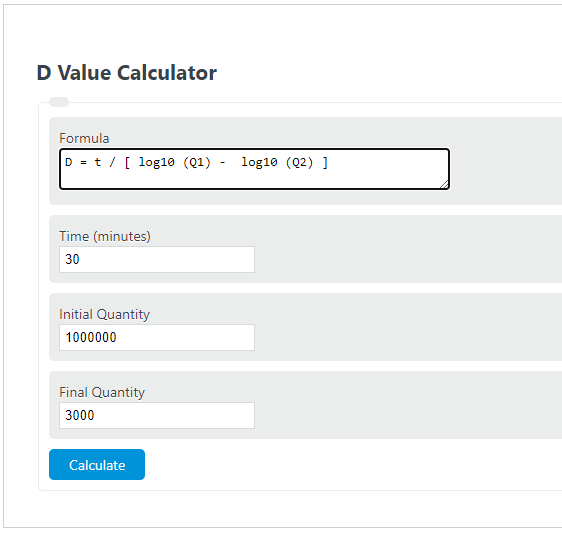
D-Value Formula
The calculator uses one of two formulas depending on the mode you select.
Find D-value from a survival experiment:
D = t / (log10(N0) - log10(N))
Time for a given log reduction when D is known:
t = D * (log reductions)
- D = decimal reduction time (time to kill 90% of the population at a fixed temperature or dose)
- t = exposure time
- N0 = initial viable count (CFU)
- N = surviving count after exposure (CFU)
- log reductions = log10(N0/N)
Assumes first-order (log-linear) inactivation kinetics at a constant temperature, pH, water activity, and treatment intensity. Real survival curves can show shoulders or tails. If conditions change, D changes too. Counts must be greater than zero. The “time for log reduction” mode does not include come-up time or lag.
Reference Values
Typical D-values for foodborne organisms at common process temperatures. Use these as ballpark figures only. Actual D-values depend on strain, substrate, and method.
| Organism | Temp | D-value |
|---|---|---|
| Salmonella spp. | 60 C | 2 to 6 min |
| E. coli O157:H7 | 60 C | 0.4 to 0.8 min |
| Listeria monocytogenes | 60 C | 2 to 3 min |
| Staphylococcus aureus | 60 C | 2 to 6 min |
| C. botulinum (proteolytic spores) | 121 C | 0.2 min (D121 reference) |
| Geobacillus stearothermophilus spores | 121 C | 1.5 to 3 min |
How log reductions translate to percent kill:
| Log reduction | Percent killed | Typical use |
|---|---|---|
| 3-log | 99.9% | Sanitizer claim |
| 5-log | 99.999% | Juice HACCP rule |
| 6-log | 99.9999% | Milk pasteurization target |
| 12-log | 99.9999999999% | Botulinum cook for low-acid canned foods |
Worked Example
A heat-resistance trial starts with 1,000,000 CFU/mL and drops to 100 CFU/mL after 8 minutes at 60 C.
log10(1,000,000) – log10(100) = 6 – 2 = 4 log reductions
D = 8 min / 4 = 2 min
To get a 6-log reduction at the same temperature: t = 2 min x 6 = 12 min.
FAQ
What is the difference between D and Z? D is the time for a 1-log kill at a fixed temperature. Z is the temperature change needed to shift D by a factor of 10.
Can D-value be less than a second? Yes. UV, high-pressure processing, and high-temperature short-time systems often have D-values in seconds or fractions of a second.
Why must final count be less than initial count? D describes inactivation. If the population grew or stayed flat, the log-linear model does not apply.
Does D-value depend on initial count? Under ideal log-linear kinetics, no. In practice, very high or very low starting populations can shift apparent D due to clumping or detection limits.