Calculate equilibrium constants Kc or Kp from concentrations or pressures, and estimate pavement k-value from CBR or resilient modulus.
- All Construction Calculators
- Rate Constant Calculator
- Molar Volume Calculator
- Equilibrium Constant Calculator
- Henry’s Law Constant Calculator
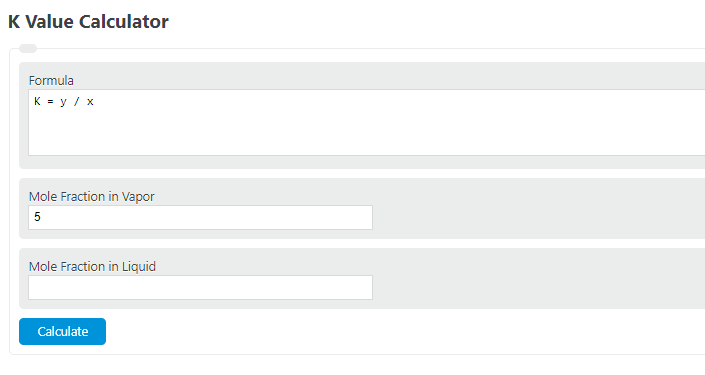
K Value Formula
The following formula is used to calculate a k value.
K = y / x
- Where K is the K value, or vapor-liquid equilibrium ratio.
- y is the mole fraction in the vapor
- x is the mole fraction in the liquid
To calculate a k-value, divide the mole fraction in the vapor by the mole fraction in the liquid.
K Value Definition
The K value is most often called the vapor-liquid equilibrium constant. It's a ratio of the mole fraction in vapor to the mole fraction in a liquid of vapor to liquid transition or state. This value is derived from Henry's Law and is sometimes called Henry's constant.
Can K values be negative?
In chemistry, as the formula above is used for, k values cannot be negative since you cannot have a negative mole fraction of a substance.
K Value Example
how to calculate k value?
- First, determine the mole fraction in the vapor.
Determine the mole fraction of the molecule in a vapor state.
- Next, determine the mole fraction in the liquid.
Calculate the mole fraction of the molecule in a liquid state.
- Finally, calculate the K Value.
Calculate the K value using the formula above.
FAQ
A mole fraction is the ratio of the number of moles of one component of a solution to the total moles of all components.