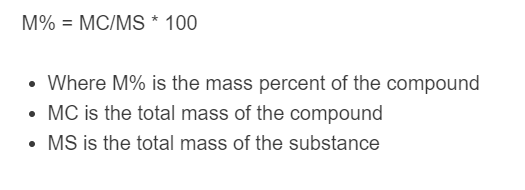Enter the mass of the component and the total mass of the sample into the calculator. The calculator will determine the mass percent of the substance. This calculator can also determine either mass when you enter the mass percent and the other mass.
Mass Percent Formula
The following equation is used to calculate the mass percent of a compound in a substance.
MP = (MC/MS) * 100
- Where MP is the mass percent of the compound
- MC is the mass of the compound (solute/component) in the sample
- MS is the total mass of the sample/solution (solute + everything else)
To calculate a mass percent, divide the mass of the component (solute) by the total mass of the sample/solution, then multiply by 100.
Mass Percent Definition
A mass percent is the mass of a component divided by the total mass of the mixture or solution, expressed as a percentage.
Mass Percent Example
How to calculate mass percent?
First, measure the mass of the compound. This should be only the mass of the compound/material you are trying to calculate the mass percent for. Use an accurate scale or balance for this measurement.
For this example, the mass of the compound is measured to be 50 kg.
Next, measure the mass of the entire substance. This is the mass of the entire solution or substance including the compound being analyzed. The same measurement techniques as step 1 should be used.
For this example, the mass of the entire solution/substance is found to be 150 kg.
Finally, calculate the mass percent using the values of the compound and substance found above.
M% = (MC/MS) * 100
M% = (50 kg / 150 kg) * 100
M% = 33.333…% ≈ 33.33%
FAQ
Mass percent is defined as the total percentage of mass that one single compound makes up out of the total mass of a solution of a substance that the compound is contained in.
For a physical sample, you measure mass using a balance/scale (and, if needed, by difference using a container). If you are instead looking for the compound’s molar (formula) mass from its chemical formula, you calculate it by summing the atomic masses of the elements in the formula.