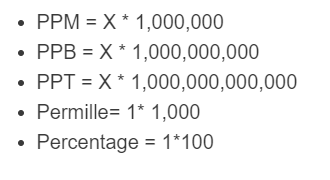Calculate PPM, PPB, PPT, percent, and mg/L conversions, or find gas concentration from molecular weight, temperature, and pressure.
- All Unit Converters
- mcg to PPM Calculator
- PPM to mg/L Calculator
- PPM to Molarity Calculator
- PPB Parts Per Billion Calculator
- Parts Per Trillion Calculator
- Percentage Calculator
PPM Formula
The following formulas are used to calculate PPM and other values, where X is a unitless fraction (ratio), such as a mass fraction or mole fraction.
- PPM = X * 1,000,000
- PPB = X * 1,000,000,000
- PPT = X * 1,000,000,000,000
- Permille = X * 1,000
- Percentage = X * 100
To calculate PPM, multiply the fractional value (X) by 1 million (1,000,000). To calculate PPB, multiply X by 1 billion. To calculate PPT, multiply X by 1 trillion.
PPM Definition
PPM is short for parts per million. It is a way of expressing a very small ratio: 1 ppm = 1 part per 1,000,000 parts = 10-6 (dimensionless). In practice, you should specify the basis (for example, mass fraction, mole fraction, or volume fraction).
For instance, if a substance makes up a fraction of 0.00005 (5×10-5) of a mixture on a stated basis (such as mass fraction), that is 0.00005 × 1,000,000 = 50 ppm.
How to calculate PPM
First, determine the ratio you want to express in parts per million. Commonly this is a mass fraction (for example, mg of solute per kg of solution), and for dilute aqueous solutions ppm by mass is often approximated from mg/L using the solution density.
For example, you might measure the mass of a substance per mass of soil (mg/kg).
Second, convert that ratio to a unitless fraction (X) if it is not already, and multiply X by 1,000,000.
This can be done using the calculator above, or it can be done using a standard calculator or hand calculation.
Finally, you need to analyze the results.
PPM is used to bring a really small number into a more realistic number that you can visualize and analyze. For instance, if your number is still low, switch to parts per billion and so on.
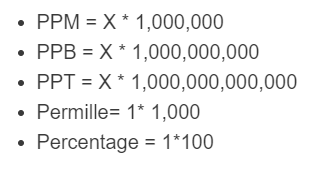
Parts-per Notation
PPM is one of a set of dimensionless quantities used to describe small ratios, as stated above.
The others—PPB, PPT, and even PPQ (parts per quadrillion)—are also part of this notation, often referred to as parts-per notation.
Although these values are dimensionless ratios, the symbols (ppm, ppb, etc.) are not SI units. They are commonly used alongside SI units, but the intended basis (mass fraction, mole fraction, etc.) should be made clear.
The most common use for parts-per notation is in chemistry. It's used to both describe and explain the abundance of materials in a solution.
Another common use is in environmental engineering. It's used to describe dissolved pollutants in water. For example, 1 mg of a pollutant per 1 kg of water is 1 ppm by mass (and for water with density near 1 kg/L, this is approximately 1 mg/L).
Parts-per notation isn't just used in chemistry, it's also used in physics. Its use in physics is tied to proportional values or phenomena.
That is, a length ratio of 1 micrometer per 1 meter can be expressed as 1 ppm, since 1 µm = 10-6 m.